Glioblastoma
Glioblastoma is the most common and aggressive primary brain tumor, and prognosis remains extremely poor. Median survival for newly diagnosed patients with best available care is approximately 14.6 months.4,5 By 2030, the incidence is expected to reach roughly 18.5K cases in the United States and 70K cases across the eight major markets.
Standard of care for newly-diagnosed glioblastoma patients includes maximal surgical resection to the extent that it is safely feasible, followed by concurrent temozolomide (TMZ) and chemoradiotherapy, and then maintenance TMZ.6
TMZ confers only a modest improvement in overall survival, extending median survival by 2.5 months over median survival of 12.1 months with surgical resection and radiotherapy alone. Notably, the meaningful benefit is largely confined to the ~30% of patients with MGMT promoter methylation. Despite overall improved survival, patients will recur or progress, most within less than six months. Since TMZ’s FDA approval in 2005, no additional glioblastoma specific drugs have been approved for the disease. For recurrent glioblastoma, treatment options are scarce, individualized, and with no universally accepted standard of care.
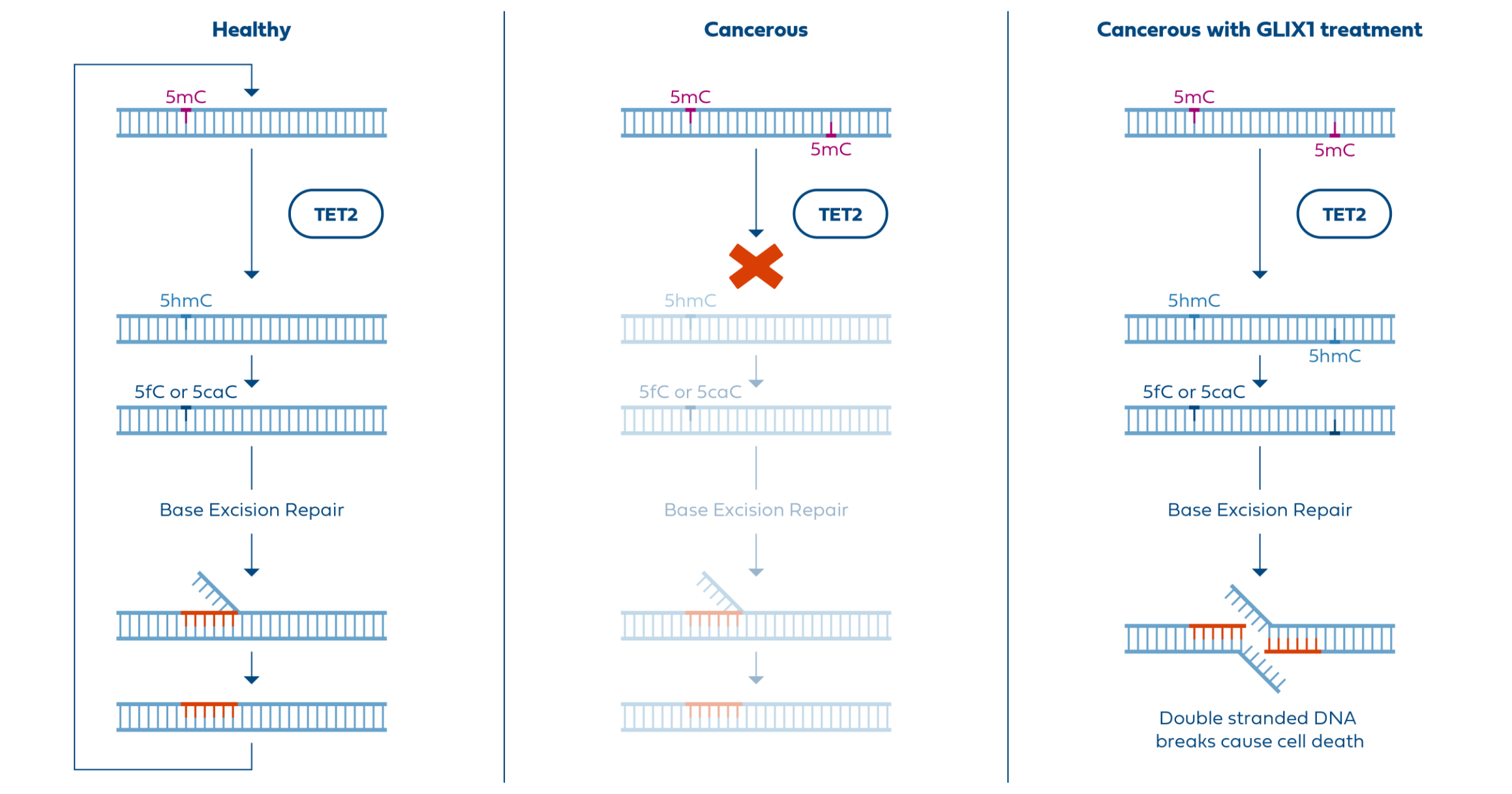
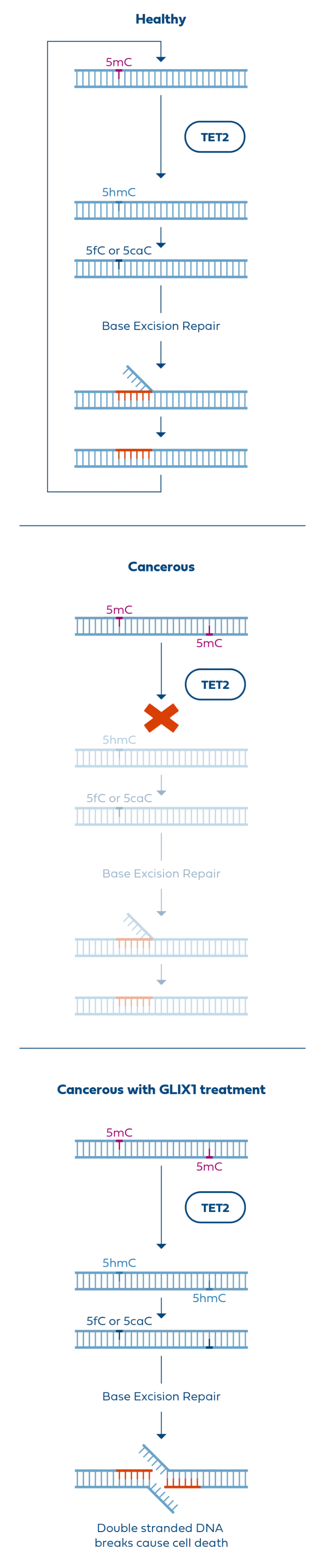
In addition to its unique mechanism, cancer-selective activity, and blood-brain penetration, GLIX1 shows efficacy in TMZ-resistant cell lines. Since most glioblastomas are initially TMZ-resistant or eventually develop TMZ resistance, GLIX1 could meaningfully expand options for this currently untreatable population and hopefully change the prognosis for this serious unmet need.
The GLIX1 first-in-human phase 1 trial is planned to recruit up to 30 recurrent glioblastoma patients. The objective is to establish a maximum tolerated dose and/or a recommended dose based on safety, PK, and preliminary efficacy. Data from the phase 1 trial is anticipated in H1 2027.
Potential cohorts for a phase 2 expansion trial could include:
- GBM – newly diagnosed and/or recurrent
- Additional cancers – with/without PARP inhibitor (PARPi) combination